What’s the 411 on the state of kidney care
in this country?
By Elise Wilfinger
05.09.2023

Introduction
As Spring approaches, we’re excited to continue our exploration of advances in kidney care and management and to shine a light on a disease which is still largely unrecognized and not prioritized.
We’re fortunate that Dr. Katherine Tuttle agreed to join us for this discussion, reprising one that we had back in August of 2021.
By way of introduction, Dr. Tuttle is a Professor of Medicine in Nephrology at the University of Washington, The Executive Director for Research at Providence Inland Northwest Health, and the Regional Principal Investigator at the Institute for Translational Health Sciences.
An internist, with three fellowships (Metabolism, Endocrinology and Nephrology), Dr Tuttle’s major research interests are in the areas of clinical and translational science and precision medicine for diabetic kidney disease, chronic kidney disease, diabetes, and hypertension.
She has served on the Board of Directors for the Kidney Health Initiative and has chaired numerous kidney and diabetes related working groups and committees including the Diabetic Kidney Disease Collaborative Task Force for the American Society of Nephrology.
At the forefront of clinical trials, Dr Tuttle has also helped deliver breakthrough therapies to prevent kidney failure and cardiovascular events.
For her considerable achievements, she’s received multiple awards, including from both the American Association of Kidney Patients and the National Kidney Foundation.
“Pain gets a person’s attention.”
- Dr. Tuttle
- Let’s start with establishing some context around the state of chronic kidney disease and why it’s often called a “silent killer.”
Chronic Kidney Disease (CKD) is a condition where people typically don’t have symptoms until late in the course. It’s hard for patients to prioritize their kidney health if they don’t even know that they have a problem. Other health conditions are noticeable, often because ‘pain’ is involved. Pain will get a person’s attention and he/she will do something about it. Even though kidney disease is more serious than many other painful, health conditions, kidneys are ‘out of sight and out of mind.’
“It’s our moral and ethical obligation.”
- Dr. Tuttle
- In your opinion, what are some of the best ways the healthcare ecosystem can band together and break this silence?
It’s important to have far-reaching vocal advocacy for changes in health policy, for the building of disease awareness, and for national education efforts. Awareness and education must be dual-focused: the professional community and the public.
CKD has a higher annual mortality rate than many oft-discussed conditions, like lung cancer and breast cancer. Like CKD, these more common cancers are also ‘silent’ until later-stage disease. What’s the difference? The Cancer community has done a remarkable job educating the public, encouraging people, especially those at high risk, to get screened. That’s because if detected early, patients have a much greater chance of survival.
We need to do the same thing for kidney disease.
We should use the Cancer outreach approach (consistent funding for advertising, public announcements, etc.) as a benchmark for how to build awareness and action for early identification and treatment for a disease that’s silent upstream but highly fatal in later stages.
I think that the onus is on the nephrology community to get the word out.
It’s our moral and ethical obligation.
Unfortunately, CKD is still very common. And among the common chronic diseases, it’s still the least recognized, the least treated and the consequences are often dire for patients who develop it. We now have treatments, that if initiated early, can save kidneys, hearts, and lives.
“Now this…is a landmark study.”
- Dr. Tuttle
- In an October 2022 issue of The New England Journal of Medicine, an article was published with results from a Study you led with several colleagues, including Dr. Keith Norris from UCLA, who was also recently featured in our blog and in a series of videos. The Study was looking at the incidence of chronic kidney disease in adult patients with diabetes. Can you share the highlights of that Study and your findings?
It’s a landmark study and I’m happy to share our findings.
First, I think it’s important to understand the difference between incidence and prevalence of a disease.
Prevalence is how many people in a population have a condition. This is relatively easy to count because it’s a single point in time. So, for example, among people known to have type 2 diabetes (T2D), we can examine electronic health records (EHR) and use eGFR and UACR results to estimate the total number of people who also have CKD.
And we know that about 40% of people with type 2 diabetes will develop CKD. That’s a lot of people, especially when we consider that the number of people with type 2 diabetes is escalating.
On the flip side, we haven’t had reliable data nor estimates on where the disease starts and its incidence, the proportion or rate of those who will develop new onset CKD during a particular period. We need to understand who will develop it, how old they are, and if there are any other discerning characteristics (gender, race, etc.).
The important part of the paper, Incidence of Chronic Kidney Disease among Adults with Diabetes, 2015-2020 is that it defines where CKD starts in diabetes. Imagine how much more success we would have in mitigating CKD by leveraging this new data in combination with awareness, education, and screening efforts.
- What patient population was the Study based on?
The Paper was based on patients in our large CURE-CKD registry.
We have the enormous capability of looking at large populations within our two healthcare systems: Providence Health and UCLA. Although we have people on the registry who have been treated since 2006, our published analysis in NEJM focused on 2015 – 2020 to provide a more contemporary dataset. In this Study, we evaluated over 650,000 people with diabetes and with no diagnosed kidney disease at baseline. We followed this subgroup of patients, gathering lab studies (eGFR and UACR) in accordance with KDIGO (Kidney Disease Improving Global Outcomes) guidelines. We required persistent measurement, at least two eGFR and UACR readings, at least 90 days apart, to ensure that the results were an accurate reflection of kidney status, not an aberration.
We found that all non-white races (except for the Asian race), have substantially higher rates of developing chronic kidney disease as compared to white Americans.
- Hawaiians or Pacific Islanders have a 60% greater incidence (than the white population) for CKD onset.
- African Americans, 40% greater.
- American Indians, 33% greater.
- Hispanics or Latinos, 25% greater.
Even though these patients are being treated in the same healthcare systems, and technically, have the same access to care, there are big disparities by race and ethnicity. Obviously, something is going on in the system.
We also found that CKD onset was more common in men than women, and that older patients (60+ years old) were at the highest risk (40-60% greater).
Looking at sequential, two-year intervals, we saw a downward trend across all groups, though some groups still had remarkably high rates for developing CKD, suggesting that overall, more than 1/3 of contemporary patients with diabetes would develop CKD over a 5-year period.
- I’m interested in your thoughts about “therapeutic inertia” in type 2 diabetes as it relates to the development of cardiorenal and metabolic diseases. Why does this inertia exist? And how do you think that we can change this at the primary care level?
It’s all about priorities; other conditions are still deemed more important.
Within CURE-CKD, we can look at medication use, for insights. In 2017, only 40% of patients with diabetes and CKD were prescribed angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs). More recently, the rate of prescribing has increased by approximately 70%. That’s good news. But if we can’t even get these baseline medications prescribed more regularly, what are the chances of adding newer therapies? Once again, it comes back to education. Both providers and patients need to understand how important this is, what the risks are, and that new treatment options are available and can help.
“If CKD-related tests are not flagged…
it’s unlikely that the tests will be ordered.”
- Dr. Tuttle
- Given what’s happened (or hasn’t happened) thus far with awareness-building and education efforts, I’m wondering if we need to change from the current provider-driven strategy to a patient-driven strategy, inspiring them to raise their hands for testing and kidney care?
I understand that perspective, but we still need to focus on both. We need a lot of voices. Providers and members of the clinical staff, patients, patient peers, family members, advocacy groups (like American Association of Kidney Patients and the National Kidney Foundation), and innovators. We also need changes to health policy.
Of course, changes to the EHR would be beneficial, as it’s on the front line. For example, there are ‘score cards’ for executing cancer screenings. As a provider, if you successfully complete all the suggested screenings, you’re rewarded. If you don’t, there is a demerit of some kind. This also applies to cardiovascular screening and vaccinations. Nothing like this exists for CKD. If CKD-related tests are not flagged, and if there’s not a related score card, it’s unlikely that the tests will be ordered.
In addition to prompting for important kidney health test ordering, we also need to provide specific care pathways and tiered workflows for how-to properly implement the Guidelines. For example:
- Once a patient is prescribed an ACE or ARB, when should they be seen next?
- When should span class=’lining_numbers’>SGLT2 inhibitor be added?
- What should ongoing monitoring look like?
- When should finerenone and the GLP-1s be considered?
If a tiered approach does not take place, we may be losing out on the opportunity for the patient to have an optimized plan for progression mitigation.
Part of the reason for ‘therapeutic inertia’ is that providers don’t always know what to do next and when to do it. The heart failure guidelines do a good job here. They support a pillar approach, providing guidance for the sequence and timing of the five available drugs (considered to be the best treatment medications) in the toolkit. This should serve as a potential model for therapeutic management in CKD, which now has four medications in its arsenal.
“CKD should no longer be a subset of diabetes.
It needs to be a standalone priority.”
- Dr. Tuttle
- What can governmental agencies do better?
CKD needs to be a standalone priority.
Today, the Centers for Disease Control and Prevention (CDC) ‘bury’ kidney disease under the diabetes program. The problem is that as a subset or complication of something else, it is never a priority on its own.
The CDC has a cardiovascular program, a diabetes program, a cancer program, but no dedicated kidney disease program.
We need to prioritize it at the public health level by health policy. That’s the way we’ll get ‘all the way to bright’ for change.
- Can you comment on the relative importance of continuous glucose monitoring for patients with advanced chronic kidney disease? Is there a role for this in early-stage patients at high risk for progression?
The foundation of good diabetes care is excellent glycemic control. In studies that go back some forty years, we’ve learned that early, intensive glycemic control in diabetes patients can almost completely prevent microvascular complications.
These monitors have been transformative for glycemic control. They help patients act according to their real-time numbers vs waiting for their next meal and finger stick. If a patient’s numbers are going down, he/she can still catch it before it’s too low, averting hyperglycemia.
Once CKD develops, glycemic control is less effective at preventing its progression. That said, it’s still important to have good glycemic control to ensure the health of the eyes, nerves, and feet.
Big picture: using continuous glucose monitoring is yet another important tool for holistic care management.
“We must make patients care about their kidneys.
We’ve done it for other organs.”
- Dr. Tuttle
- Since CKD is largely asymptomatic in its early stages, how can we get patients to even care about their kidneys and engage in treatment?
Again, we need to build awareness and educate to ‘make them care.’
Public health campaigns, public service announcements, direct to patient advertising with inspiring mantras, direct to provider marketing, all drove staggering increases in screenings for other asymptomatic diseases like breast and cervical cancers.
Importantly, the U.S. Preventive Health Services Taskforce is reconsidering the need for CKD screening. It recommended against it about a decade ago, likely because there were not many available treatments at the time. So much has changed on that front.
If patients can understand that mammograms are important for breast health, they can also understand that screening for CKD is important, for kidney and for overall health.
“In my Clinic today, we have 578
unassigned requests for nephrology consults.”
- Dr. Tuttle
- I’m interested in what differences exist between how you and other nephrologists manage patients with early-stage chronic kidney disease?
First, and unfortunately, the state of nephrology today disincentivizes us from seeing early-stage patients. We have a dire workforce issue that forces us to focus on people with advanced kidney disease.
Here’s a tangible example, in my Clinic today, we have requests for 578 unassigned nephrology consults. And most of these patients don’t have early-stage CKD.
- If you can’t see these patients in earlier stages, are you able to help guide primary care doctors in this pursuit?
We have collaborations across departments at Providence Inland Northwest Health. That said, it would help primary care to be given a more detailed workflow approach, based on the bedrocks of our therapy.
For example:
- Give patient an ACE and ARB as a first line therapy.
- 4-6 weeks later, add a SGLT2i.
- In another 4-6 weeks, determine whether finerenome or a GLP-1 should be added, based on albuminuria, blood pressure, cardiovascular risk, and hemoglobin A1C.
PCPs would also benefit from EPIC alerts on their patient screen. To avoid ‘alert burnout,’ I don’t think that every alert should “pop up” for every patient. If a patient is deemed high risk for a certain condition, the alert would populate accordingly (i.e., order Test A).
There’s potential in using something like the Kidney Failure Risk Equation (side note: used to determine 2- and 5-year probability of treated kidney failure (dialysis or transplantation) for a patient with CKD, stages 3-5), or your company’s test (side note: KidneyIntelX™ predicts risk for rapid progressive decline in kidney function in stages 1-3b) to identify high risk patients for kidney failure. This would be a form of precision medicine. Patients would receive the screenings that they really need.
“If you were driving your car, faster and faster, it would eventually wear out.
So would your kidneys.”
- Dr. Tuttle
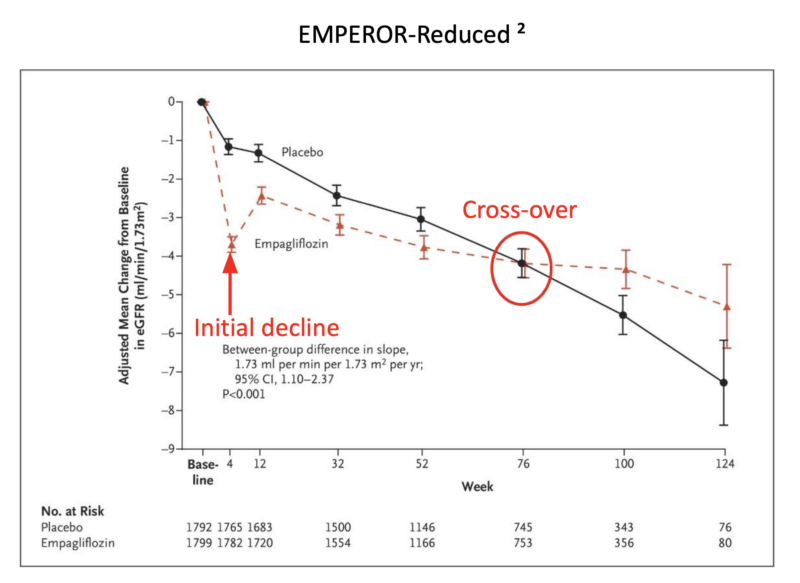
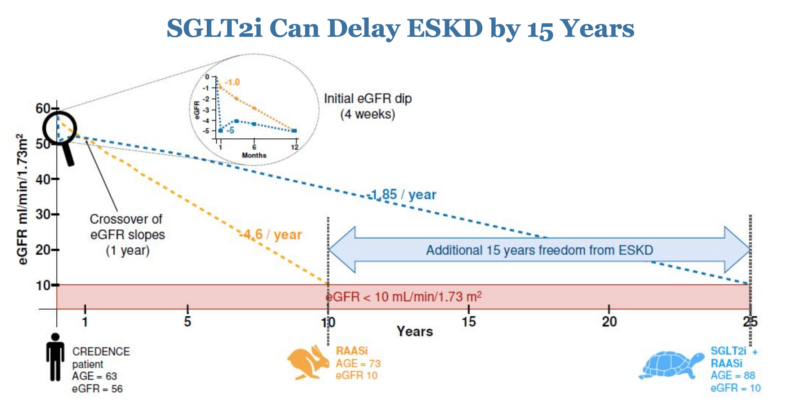
- In our last conversation, we talked about the exciting, newer class of medications named SGLT2 inhibitors (SGLT2i). We’ve heard that doctors sometimes have trouble explaining the initial downward slope that occurs when a patient takes these therapeutics. How would you suggest that a primary care physician explain it?
In progressing diabetic kidney disease, as in other forms of kidney disease, functioning kidney units or nephrons are lost over time and the remaining kidney tissue picks up the slack. This process of compensation is called hyperfiltration, and it’s associated with hypertension, which can be systemic (measured in the arm) or localized (directly in the kidney). Hypertension is very damaging to vascular structures like kidneys. And because 25% of cardiac output is in your kidneys, anything we can do to lower pressure in those blood vessels, will help them to function better.
Our toolkit of medications – ACEs, ARBs and SGLT2is – work in part by reducing this hyperfiltration, alleviating the pressure in the kidney’s filtering units.
Initially, when taking these medications, there is an abrupt decrease in filtration. The total eGFR will go down, which may be alarming to both provider and patient. But here’s what’s happening: the kidneys are moving from a hyperfiltration state down to a more normal filtration state. The medications are stopping the kidneys from running at hyper-speed. Over time, that translates to preserved kidney function.
What if we put it in ‘car’ terms. If you were driving your car, faster and faster, it would eventually wear out. Yes, in the short term, you would be going faster. Longer term, however, you’ll likely kill your car.
If you look at the slope or trajectory of kidney function over time, patient GFR numbers stabilize. The curves cross around 18 months, which is relatively early in the span of a lifetime.
- What’s happening with the Kidney Precision Medicine Project? Where are we relative to the goals of that project?
It’s going very well. We’re still in the vigorous data collection stage.
There are hundreds of kidney biopsies that have been collected in patients with CKD or acute kidney injury, attributed to diabetes or hypertension. We’re looking at biomarkers in both blood and urine along with tissue interrogation.
The goal is to better understand nuanced mechanisms at the molecular level to improve our ability to develop new, and even more effective therapies, and how to use them.
- Any last minute 411 items?
Yes, we can never forget the importance of a healthy lifestyle. Staying physically active, and maintaining a healthy diet, will likely enable a reduction in the number of medications a patient needs to take. And anyone who follows a healthier lifestyle, feels better all around.
Join the List
To be one of the first to read our upcoming blogs and other content, subscribe to Well FilteredTM here.
Spread the word
If you enjoyed this post, we’d be grateful if you would help us spread its content.